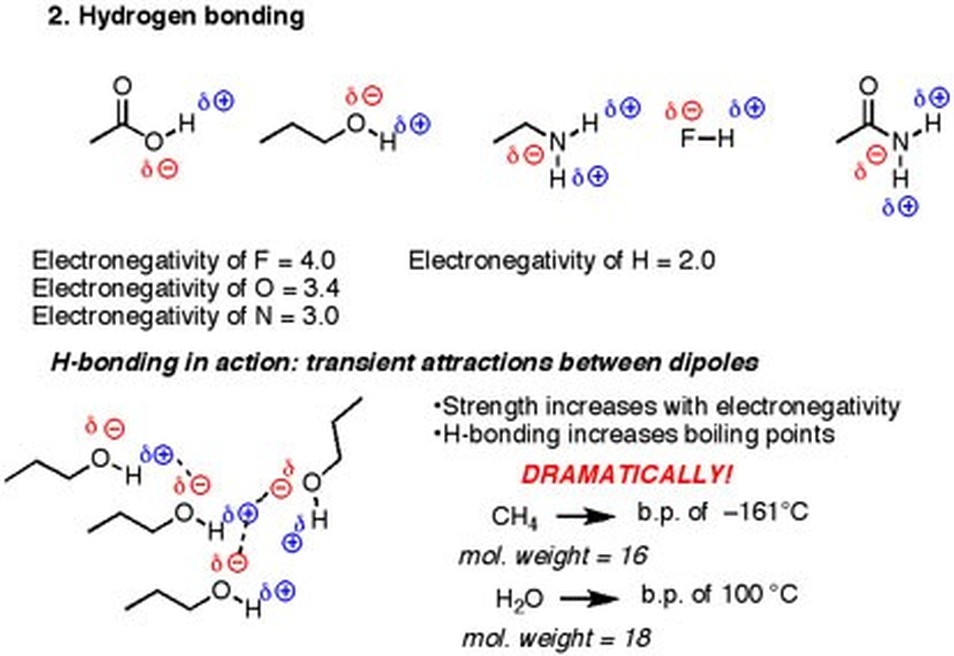
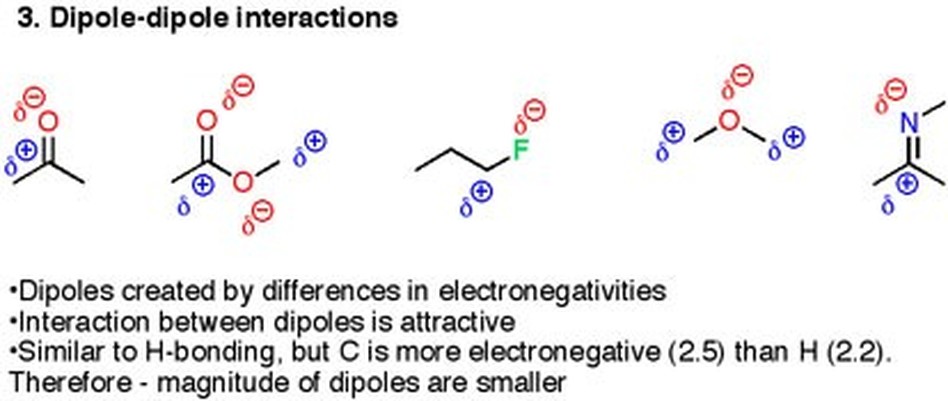
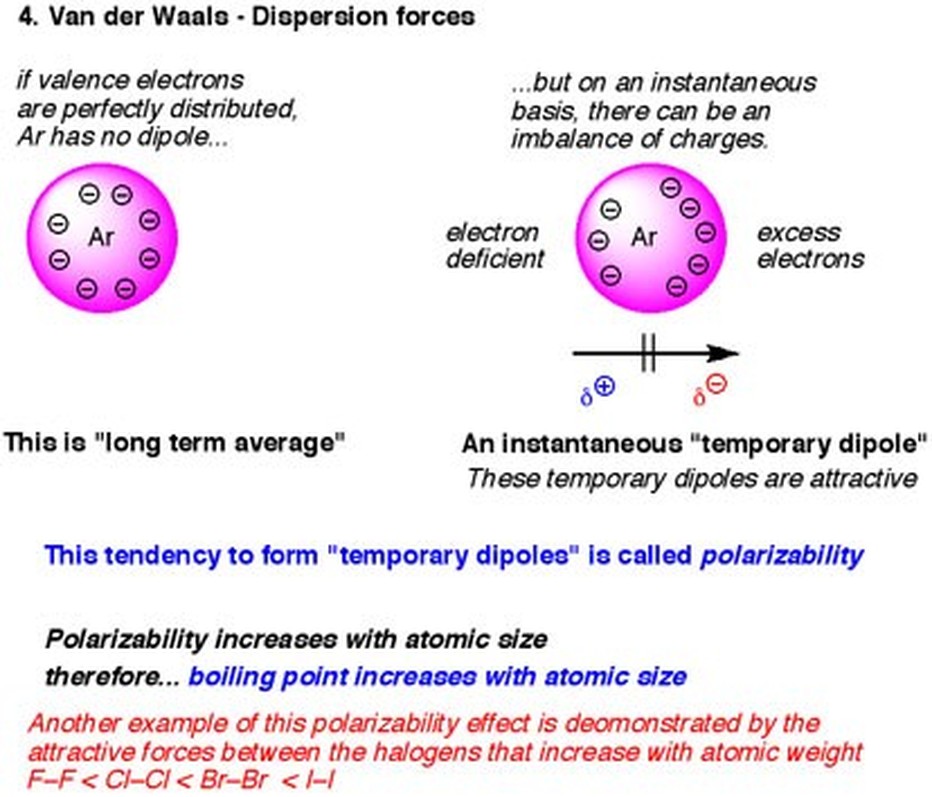
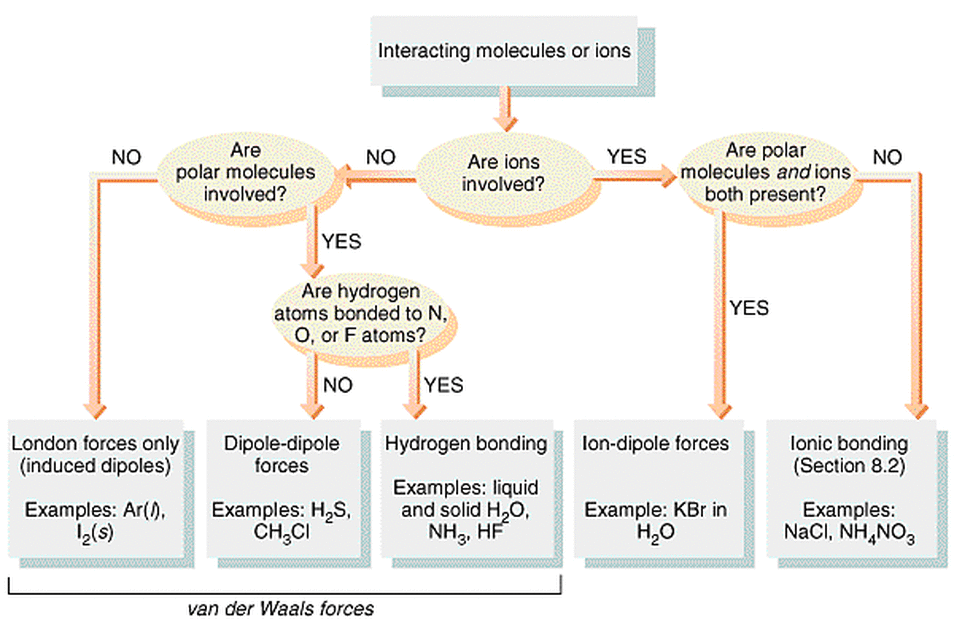
Description
The Intermolecular Forces (forces between molecules) are weaker than Intramolecular Forces (The Chemical Bonds within an Individual Molecule). This distinction is the reason we define the molecule in the first place. The properties of matter result from the properties of the individual molecule (resulting from chemical bonding) and how the molecules act collectively (resulting from intermolecular forces).